Expert Seminar on the Internationalization of Doctoral Programs

Millions of people around the world suffer from organ failure, and the demand for transplants far exceeds the number of available donor organs.
In light of this disparity, three-dimensional (3D) bioprinting is emerging as one of the most promising technologies in regenerative medicine. The prospect of producing personalized organs from a patient’s own cells could not only solve the problem of donor shortages but also eliminate the need for toxic immunosuppressive drugs, which are an integral part of traditional transplantation.
A key factor distinguishing bioprinting from traditional 3D printing is the use of so-called bioinks—special formulations containing living cells, structural proteins, and growth factors—in place of plastics, which enable the recreation of the complex architecture of biological tissues.
In medical 3D bioprinting, the starting point is a digital model of the organ created from images obtained via computed tomography or magnetic resonance imaging. Based on this, the software generates a series of very thin cross-sections, which the bioprinter reproduces sequentially by applying successive layers of material, thereby gradually building a three-dimensional structure.
The cells used in bioprinting can be obtained directly from the patient, ensuring full immunological compatibility and eliminating the risk of transplant rejection. The source of cells can be either differentiated cells collected during a biopsy or stem cells—including induced pluripotent stem cells (iPSCs), which can be generated from mature cells in the body, and then reprogrammed to differentiate into virtually any cell type.
Importantly, the design of organ models and the analysis of biological data for bioprinting are increasingly based on machine learning algorithms and bioinformatics tools—AI algorithms optimize the composition of the bioprint, monitor the printing process in real time, and predict tissue behavior after implantation. It is precisely this combination of biotechnology and artificial intelligence that opens up entirely new possibilities for regenerative medicine.
Bioinformatics—which combines molecular biology, computer science, and data analysis—provides tools for modeling genomic sequences, selecting suitable stem cells, and predicting tissue compatibility. Those interested in this field can develop their skills throughthe postgraduate program in Bioinformatics atPJAIT, which combines knowledge in biology, medicine, and computer science, or through the new Bioinformatics — a master’s degree program atPJAIT—which prepares students for research and development work at the intersection of life sciences and technology.
Modern 3D bioprinters already utilize several tissue-engineering techniques, each of which is suited to slightly different applications:
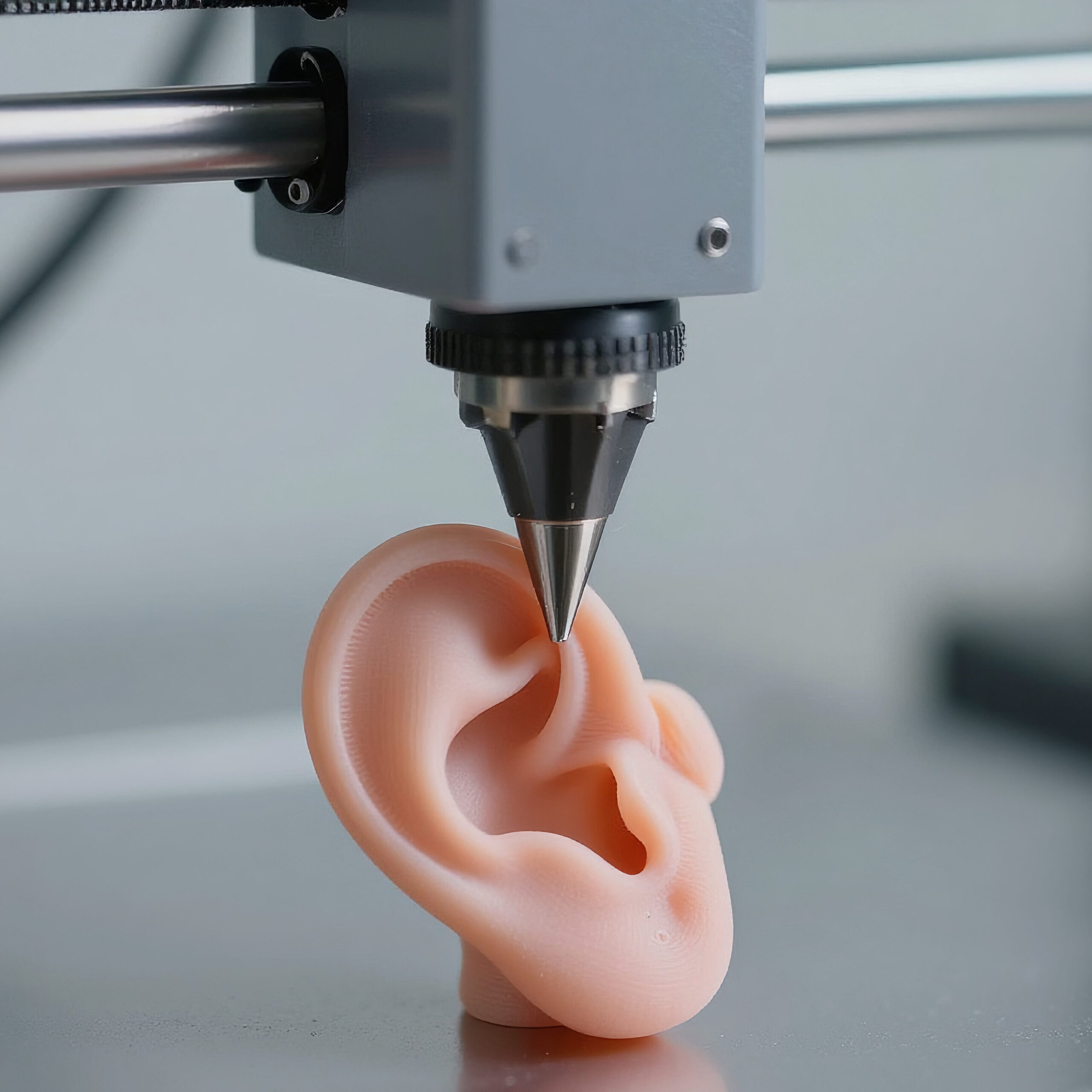
One of the most spectacular milestones in 3D bioprinting is the AuriNovo implant, developed by 3DBio Therapeutics. In 2022, the world’s first procedure to implant a 3D-printed ear was performed on a patient with microtia—a rare congenital defect characterized by the absence or underdevelopment of the auricle.
The implant was created using the patient’s own chondrocytes (cartilage cells) embedded in a collagen hydrogel, and its shape was precisely modeled based on a 3D scan of a healthy ear. The U.S. FDA has granted AuriNovo orphan drug status and designation for a rare pediatric disease, and ongoing Phase I/IIa clinical trials aim to confirm the safety and efficacy of this method.
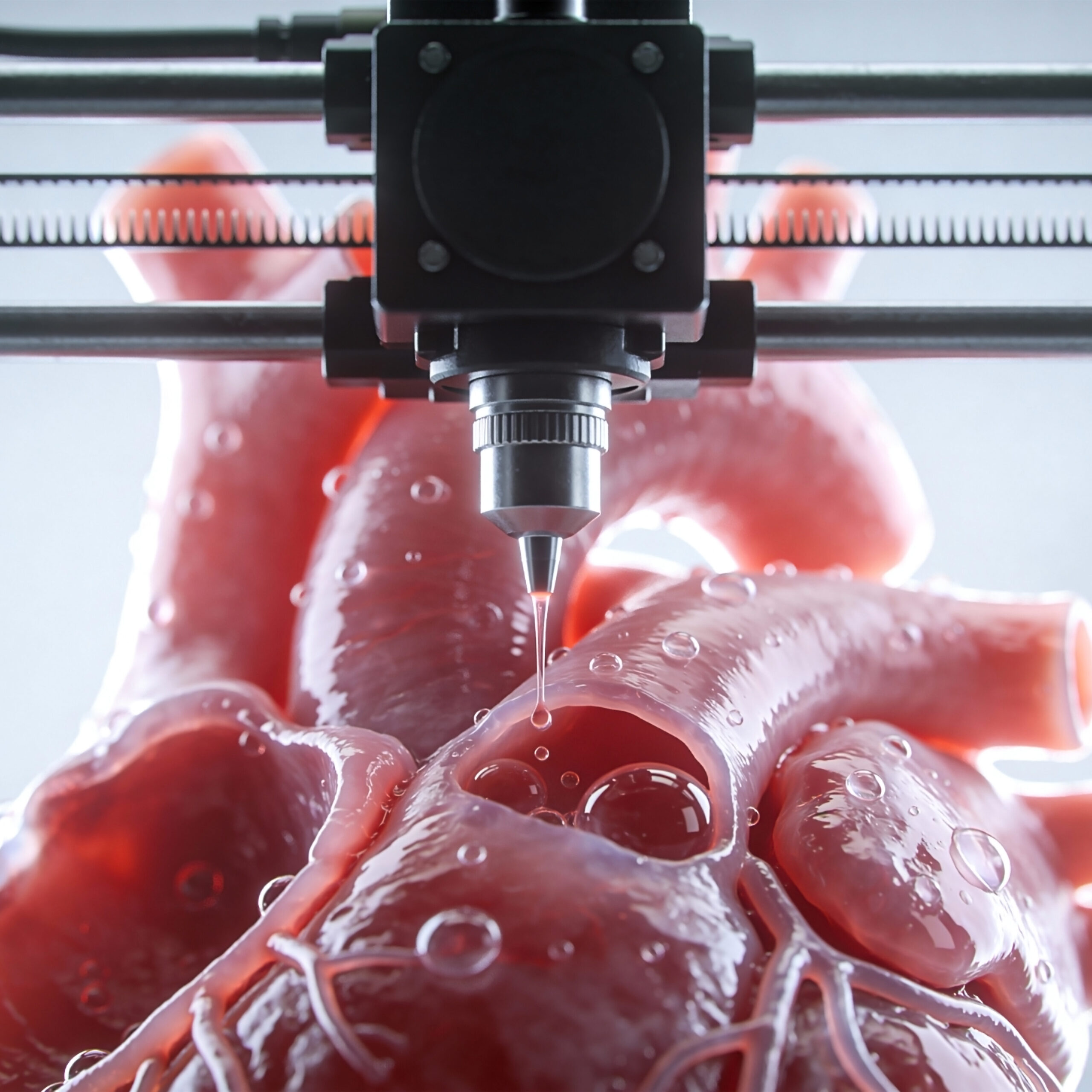
In 2019, a team led by Prof. Tal Dvir at Tel Aviv University printed the world’s first 3D-printed heart containing cells and blood vessels. The organ—about the size of a rabbit’s heart—was created using cells and biological materials taken from the patient.
The researchers obtained adipose tissue, from which they isolated cells and transformed them into cardiomyocytes and endothelial cells, and used the extracellular matrix to create a hydrogel that served as a bio-ink. Although the printed heart was able to contract, it was not yet capable of coordinated blood pumping—so the next step was to work on mastering the synchronized function of the cells and conducting tests on animal models.
In January 2026, ARPA-H (the Advanced Research Projects Agency for Health) awarded a $28.5 million grant to a team led by Carnegie Mellon University to develop a bioprinted liver for patients with acute liver failure.
As part of this LIVE (Liver Immunocompetent Volumetric Engineering) project, the FRESH 3D technique mentioned above will be used to print liver tissue composed exclusively of human cells and structural proteins, such as collagen. The engineered liver is not intended to be a permanent organ replacement—its purpose is to function for 2–4 weeks, giving the patient’s own liver time to regenerate.
What is key to this project is the fact that the liver is the only internal organ capable of regenerating on its own, which is why the "temporary bridge" strategy is particularly promising.
The biggest obstacle to bioprinting full-size organs remains the recreation of the vascular network. Reproducing the hierarchical vascular structure—from large arteries and veins, through arterioles, to microscopic capillaries—poses a fundamental engineering challenge. Without a functional vascular network, printed tissues quickly die due to hypoxia.
Printing a structure that resembles an organ is only half the battle—achieving full functional maturity is key. Cells must not only survive the printing process, but also organize themselves into proper tissue structures, establish neural connections, and begin to perform the functions specific to that organ. For example, a printed heart must be capable of coordinated beating, a kidney of filtering blood and reabsorbing substances, and a liver of metabolizing toxic substances.
In addition, scaling the technology from small laboratory models to full-size human organs poses enormous logistical challenges—such as obtaining billions of cells and maintaining their viability during the many hours of printing. Meeting these challenges requires specialists who combine biological expertise with the ability to process and analyze large biological datasets.
This is precisely the kind of interdisciplinary knowledge offered by the postgraduate program in Bioinformatics atPJAIT andfield of study within the master’s degree program —for those who wish to play an active role in shaping the future of regenerative medicine.
3D bioprinting in medicine is no longer just an experimental curiosity—it is a heavily funded, multidisciplinary field of science that is making tangible progress year after year. The road to routine on-demand organ printing is still long, but every new breakthrough—from a 3D-printed ear to the ARPA-H liver program—brings us closer to a time when no patient will have to die while waiting for a donor